
The Forefront of Tendon Injury Treatments
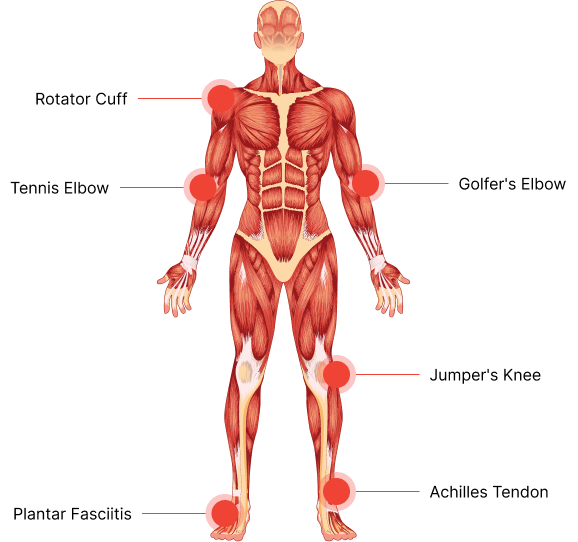
Chronic and painful disorders of tendons and ligaments, such as tendinopathy, are frequent among both active/athletic and inactive individuals. These patients are usually treated by general or sports medicine physicians, yet therapeutic options remain limited and frequently ineffective.
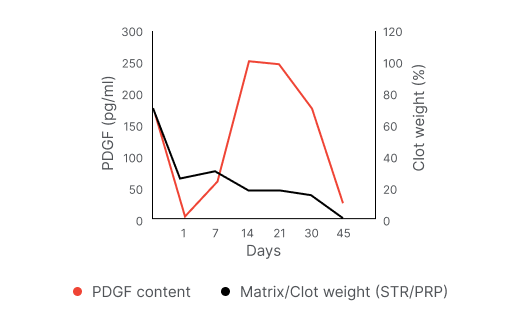
One emerging approach involves the use of autologous platelet-rich plasma (PRP) – a blood-derived concentrate rich in platelets and associated healing growth factors. However, PRP usually displays poor retention of the platelets at the injury site and their fast dissipation may limit the effect of the growth factors.
2%
The global population suffering from tendinopathy¹²³
40%
Sporting injuries involve tendon disorders⁴
>30%
Of musculoskeletal pain consultations are related to tendon injuries⁴
A Revolutionary Treatment for Tendinopathy
Vergenix™STR is intended for treatment of tendinopathy.
Vergenix™STR is a soft tissue repair matrix designed to be mixed with autologous PRP to form a scaffold with delayed degradation and enhanced retention to the injury site, in order to enable a localized and prolonged therapeutic effect.
How Vergenix™STR makes the difference?
Chronic tendinopathy is relieved
Upon injection into an injury site, the mixture of Vergenix™STR and autologous PRP forms a collagen-fibrin matrix which holds the platelets in-situ, allowing prolonged release of growth factors at the injury site.
Made from human collagen
CollPlant developed a patented process that enables to produce recombinant human collagen in plants. As a result, Vergenix™STR eliminates the risk of allergic response and exposure to animal pathogens, providing safe treatment alternative.
A single application is sufficient
Upon injection into an injury site, the mixture of Vergenix™STR and autologous PRP forms a collagen-fibrin matrix which holds the platelets in-situ, allowing prolonged release of growth factors at the injury site.
Proven in use
CollPlant developed a patented process that enables to produce recombinant human collagen in plants. As a result, Vergenix™STR eliminates the risk of allergic response and exposure to animal pathogens, providing safe treatment alternative.
Time course of growth factor release ⁴
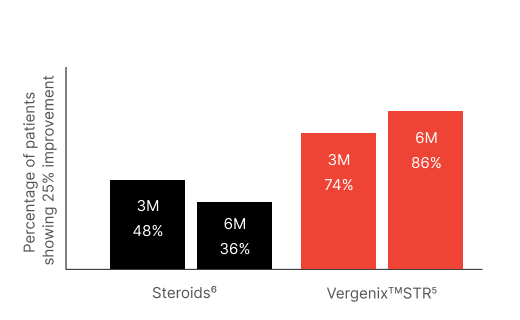
Clinical study, 40 patients
Recovery in pain and motion score (PRTEE) after treatment with Vergenix™ STR and PRP
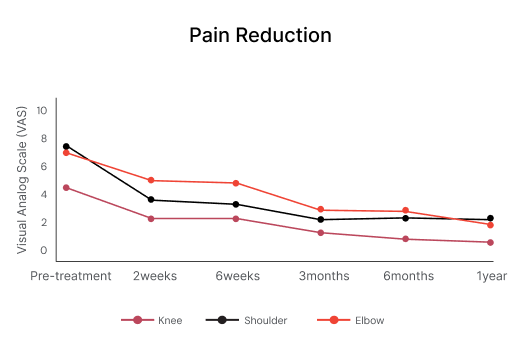
Pain score (VAS) reduction after treatment with Vergenix™ STR and PRP⁴ ⁷
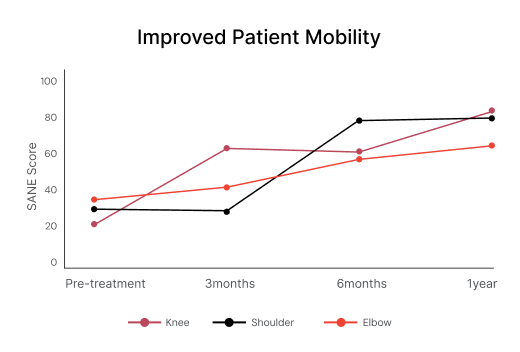
Functionality score (SANE) improvement after treatment with Vergenix™ STR and PRP⁴ ⁷
Sources:
- Iris Sophie Albers et al., Incidence and prevalence of lower extremity tendinopathy in a Dutch general practice population: a cross sectional study, BMC Musculoskeletal Disorders, 201617:16
- De Jonge S et al., Incidence of midportion Achilles tendinopathy in the general population, Br J Sports Med. 2011 Oct;45(13):1026-8
- Kelsey Lipman et al., Tendinopathy: injury, repair, and current exploration, Drug Design, Development and Therapy, 2018:12 591–603
- Christoph Elser et al., ACP Tendo-Plant Derived Human Collagen Scaffold Combined With ACP for the Treatment of Tendinopathy—A European Case Series. Poster No. 1216 presented at: ORS 2018 Annual Meeting; March 12, 2017; New Orleans, L
- Uri Farkash et al., First clinical experience with a new injectable recombinant human collagen scaffold combined with autologous platelet-rich plasma for the treatment of lateral epicondylar tendinopathy (tennis elbow). J Shoulder Elbow Surg (2018)
- Peerbooms JC et al., Positive Effect of an Autologous Platelet Concentrate in Lateral Epicondylitis in a Double-Blind Randomized Controlled Trial Platelet-Rich Plasma Versus Corticosteroid Injection With a 1-Year Follow-up. Am J Sports Med (2010)
- Clinical Outcomes Report, data for ACP Tendo provided to CollPlant through the Surgical Outcome Systems™, Arthrex, updated 2019
Learn more about Vergenix™ STR
VergenixSTR™ Brochure