rhCollagen
Collagen is an essential building block of the human body, providing structural support and biological signals to connective tissues and organs. Therefore, it is a crucial component for tissue regeneration.
rhCollagen
Collagen is an essential building block of the human body, providing structural support and biological signals to connective tissues and organs. Therefore, it is a crucial component for tissue regeneration.
The Limitations of Traditional Collagen Sources
Traditionally, collagen has been extracted from animal and human cadaver sources. These sources bear the risk of tissue remnants triggering immune response and allergies.
Harsh extraction conditions may lead to a denatured structure and impairment of cell binding domains, compromising collagen’s functionality.
Furthermore, the highly complex and stringent post-translational modification (PTM) processing of collagen has prevented developing large-scale production of recombinant human collagen.

The Limitations of Traditional Collagen Sources
Traditionally, collagen has been extracted from animal and human cadaver sources. These sources bear the risk of tissue remnants triggering immune response and allergies.
Harsh extraction conditions may lead to a denatured structure and impairment of cell binding domains, compromising collagen’s functionality.
Furthermore, the highly complex and stringent post-translational modification (PTM) processing of collagen has prevented developing large-scale production of recombinant human collagen.

The Breakthrough: Plant-Based rhCollagen
CollPlant is the first company to develop a proprietary plant-based platform to mass produce Type I recombinant human collagen (rhCollagen).
CollPlant genetically modified tobacco plants by integrating into their genome, the five human genes responsible for human Procollagen synthesis.
Procollagen is extracted from the leaves of mature plants and subsequently processed to obtain highly purified recombinant human collagen (rhCollagen), suitable for the manufacturing of medical products.

The Breakthrough: Plant-Based rhCollagen
CollPlant is the first company to develop a proprietary plant-based platform to mass produce Type I recombinant human collagen (rhCollagen).
CollPlant genetically modified tobacco plants by integrating into their genome, the five human genes responsible for human Procollagen synthesis.
Procollagen is extracted from the leaves of mature plants and subsequently processed to obtain highly purified recombinant human collagen (rhCollagen), suitable for the manufacturing of medical products.

rhCollagen – A New Gold Standard for Tissue Regeneration
Our recombinant human collagen (rhCollagen) is structurally and functionally identical to native human Type I collagen, making it an ideal foundation for regenerative medicine applications.
Compared to tissue-derived collagens, our rhCollagen offers enhanced biofunctionality, high batch-to-batch consistency, and a significantly reduced risk of immune response, positioning it as a superior alternative for advanced biomedical use.


rhCollagen – A New Gold Standard for Tissue Regeneration
Our recombinant human collagen (rhCollagen) is structurally and functionally identical to native human Type I collagen, making it an ideal foundation for regenerative medicine applications.
Compared to tissue-derived collagens, our rhCollagen offers enhanced biofunctionality, high batch-to-batch consistency, and a significantly reduced risk of immune response, positioning it as a superior alternative for advanced biomedical use.
Main Advantages
Better Bio-Functionality
- Accelerates human cell proliferation
- Faster tissue healing
Superior Homogeneity
- Controlled physical / rheological properties
- Reproducibility
Improved Safety & Greater Purity
- Non-immunogenic
- Non-allergenic
Molecular, Physical and Biological Characterization*
Production of type I human collagen is a complex process, starting at the intra-cellular level, with the synthesis of the correct mRNA, translation into the primary amino acid sequence, post translational modifications and assembly of a stable Procollagen. Conversion of the extracted Procollagen into collagen via enzymatic digestion is followed by purification.
Extensive studies done to characterize the protein at the different stages confirmed its identity to human collagen at the molecular, physical and biological levels.
Molecular, Physical and Biological Characterization*
Production of type I human collagen is a complex process, starting at the intra-cellular level, with the synthesis of the correct mRNA, translation into the primary amino acid sequence, post translational modifications and assembly of a stable Procollagen. Conversion of the extracted Procollagen into collagen via enzymatic digestion is followed by purification.
Extensive studies done to characterize the protein at the different stages confirmed its identity to human collagen at the molecular, physical and biological levels.

The following diagram illustrates the various characterization studies conducted,
demonstrating rhCollagen identity to human collagen.
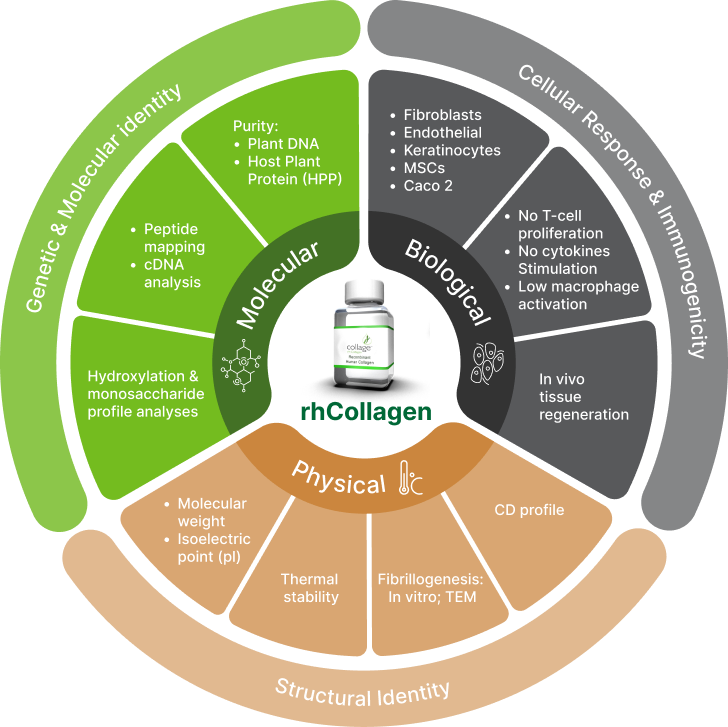
DNA inserts of the 5 genes translated into human sequence of COLα1 and COLα2 and relevant hydroxylation profile.
Secondary / tertiary structural identity of the mature collagen.
Expected functions at the structural and biological levels including fibrillogenesis, support of cells adhesion and proliferation and non-immunogenicity.
Superiority over animal & human tissue-extracted collagen in physical and biological properties.
*References:
- H.J. Seeherman et al., A BMP/Activin Chimera is superior to native BMPs and induces bone repair in non-human primates when delivered in a composite matrix. Sci Transl Med 11, eaar4953 (2019)
- Majumdar S. et al., Influence of collagen source on fibrillar architecture and properties of vitrified collagen membranes. J Biomed Mater Res Part B (2015)
- Shilo S. Cutaneous wound healing after treatment with plant-derived human recombinant collagen flowable gel. Tissue Eng Part A. 19(13-14):1519-26 (2013)
- Stein H, et al., Production of bioactive, post-translationally modified, heterotrimeric, human recombinant type-I collagen in transgenic tobacco. Biomacromolecules (2009)
- Willard JJ. et al., Plant-derived human collagen scaffolds for skin tissue engineering. Tissue Eng Part A. 19(13-14):1507-18 (2013)